 Gen info Gen info
- Sophora is a genus of about 45 species of small trees and shrubs in the pea family Fabaceae. The generic name derives from sophera, an Arabic name for a pea-flowered tree.
-
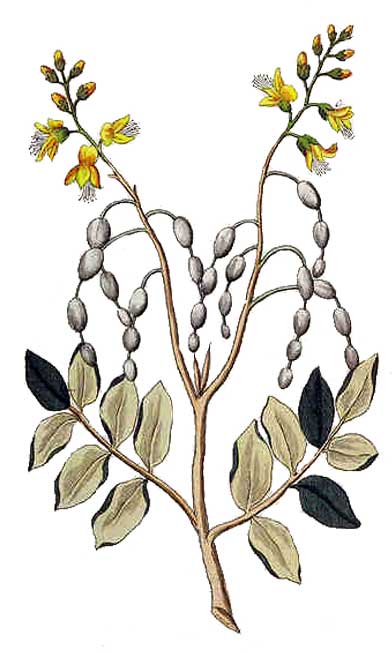
The common name Necklacepod derives from the characteristic string of seed pods that develop after the yellow flowers germinate into seeds.
-
Etymology: The genus Sophora is an Arabic name for a pea-flowered tree. The species epithet derives from Latin tomentosa meaning 'thickly matted with hair', possibly referring to the underside of leaves. (20)
Botany
Tambalisa is a gray, hairy shrub. Leaves are alternate, pinnately compound, about 15 to 30 centimeters in length, with 11 to 17 leaflets which are oval, 2.5 to 4 centimeters in length and extremely hairy. Flowers are bright yellow, about 1 centimeter long, borne on terminal racemes about 15 centimeters long. Pods are 10 to 15 centimeters in length, containing 6 to 8 seeds. Pods are characteristically rounded where the seeds occur, very narrow in the portions between the seeds.
 Distribution Distribution
- Abundant along the seashore throughout the Philippines.
- Pantropic strand plant.
Constituents
- Contains an alkaloid, sophorine, identical to cytisine from Cytisus laburnum and ulexine from Ulex europaeus. Cytisine resembles nicotine in its action.
- Study yielded a thick, red-brown fluid from the seeds, physiologically resembling the alkaloid cytisine from C laburnum seeds. Study also showed both ulexine and sophorine to be identical to cytisine. (Q)
- Study isolated two flavonoid compounds, sophoraisoflavanone A and sophoraflavanone B, together with sophoronol, isosophoranone and isobavachin from the aerial parts. (see study below) (12)
- Studies of acetone extract of dried aerial parts yielded two pterocarpans, sophoracarpans A and B, with three known isoflavonoids, wighteone (erythrinin B), sophoraisoflavanone A and I-maackianin. (1)
- Study of fresh leaves yielded three lupin alkaloids: (−)-epilamprolobine, (+)-epilamprolobine N-oxide and 5-(3′-methoxycarbonylbutyroyl)aminomethyl-trans-quinolizidine N-oxide, along with (+)-matrine, (+)-matrine N-oxide, (+)-sophocarpine N-oxide, (−)-anagyrine, (−)- baptifoline, (−)-cytisine, (−)-N-methylcytisine, (−)-N-formylcytisine, (−)-N-acetylcytisine and (±)-ammodendrine. (3)
 - Study of aerial parts of Sophora tomentosa isolated two new benzofuran derivatives (1), mp 235 - 2370, C15H10O5 and (2), mp 179 - 1810, C16H12O5, together with 1-maackiain, stigmasterol, medicagol, formononetin and 2',4',4-trihydroxychalone (isoliquiritigenin). (8) - Study of aerial parts of Sophora tomentosa isolated two new benzofuran derivatives (1), mp 235 - 2370, C15H10O5 and (2), mp 179 - 1810, C16H12O5, together with 1-maackiain, stigmasterol, medicagol, formononetin and 2',4',4-trihydroxychalone (isoliquiritigenin). (8)
- Study of root and stem of S. tomentosa isolated five new flavonoids, Tomentosanols A-E, along with 15 known flavonoids. (9)
- GC/MS analysis of S, tomentosa leaves yielded 87.74% for unsaponifiable matter and 38.70% for fatty acid methyl esters. Dominant compounds in unsaponifiable matter were n-nonacosane 43.80% and 2-methyltriacontaine 11.94%. In saponifiable fraction, saturated fatty acids were 58.37%, with 34.51% unsaturated fatty acids. Major compounds was methyl palmitate 40.02%. (see study below) (13)
- Study isolated two new pterocarpans named sophoracarpans A and B, elucidated as 6β, 9-dimethoxy-3-hydroxyperocarpan (I) and 3-hydroxy-6β-methoxy-8, 9-methylenedioxypterocarpan (III). (15)
- GC/MS analysis of essential oils from fresh flowers identified 17 components (85.08%) with major components of n-pentacosane (36.83%) and n-heptacosane (19.03%).
(17)
- Study of roots isolated a new chromone, named sophorachromone A (I), mp 138-139°C, C19H22O4, and a new flavanone, named sophoraflavanone A (II), mp 144-145°C, C25H28O5. The structures of I and II were established to be 8-geranyl-5, 7-dihydroxychromone and 6-geranyl-5, 7, 4'-trihydroxyflavanone, respectively, on the basis of chemical and spectral evidence. (19)
 Properties Properties
- Roots are yellow.
- Bark is bitter; the seeds more so.
- Plant considered diuretic, stomachic, sudorific, purgative, febrifuge.
- Seeds considered anticholeric, purgative, febrifuge, stomachic.
- Seeds are dangerously emetocathartic due to the cytisine-like alkaloid that is released.
- Studies have suggested antifungal, antimicrobial, radical scavenging, anti-parkinsonism, neuroprotective, anti-SARS-CoV-1, Anti-MERS-CoV, insecticidal properties.
 Parts used Parts used
Oil, roots, stems, seeds.
Uses
Edibility
- The pod is inedible.
Folkloric
- In the Philippines, plant used as a common remedy for stomach disorders; seeds used for stomach affections.
- Decoction of root, stem or seeds considered anticholeric. Seeds considered purgative; as little as two seeds may provide a drastic purgative action.
- Seeds used as febrifuge and stomachic.
- Oil from seeds applied externally for bones aches associated with colds. Also, used as expectorant.
- In New South Wales, seeds are used for bilious sickness.
- In Eastern Malaysia, plant used for cholera and diarrhea; also, as antidote for eating poisonous fish and other marine animals.
- Powdered seeds used by the Malays as astringent for diarrhea; leaves and roots also so employed.
- In India, seeds and infusion of roots used to treat bilious attacks.
- In Fiji, crushed leaves in coconut oil use for treatment of fractures. (10)
Studies
• Pterocarpans / Wigteone / Antifungal: Studies yielded two pterocarpans, sophoracarpans A and B, with three known isoflavonoids, wighteone (erythrinin B), sophoraisoflavanone A and I-maackianin. Wighteone is a known antifungal phytoalexin in several genera of leguminous plants. (1)
• Phenylflavanones: Prenylflavanones from S tomentosa exhibited tumor-specific cytotoxicity and antimicrobial activity.
• Phenylflavanones / Radical Scavenging / Antimicrobial / Cytotoxic: Study was done on the biologic activity of ten phenylflavanones purified from S tomentosa and S moorcroftiana. Some of the flavanones showed tumor-specific cytotoxic activity, antimicrobial activity, and anti-HIV activity, radical generation, and scavenging activity. (2)
• Antifungal / Aerial Parts: Study isolated two flavonoid compounds, sophoraisoflavanone A and sophoraflavanone B, together with sophoronol, isosophoranone and isobavachin from the aerial parts. Sophoraisoflavanone A (1) exhibited antifungal activity. (12)
• Antimicrobial / Cytotoxic / Leaves: Study evaluated phytoconstituent composition, antimicrobial, and cytotoxic activities of lipoidal matter of leaves of S. tomentosa and S. secundiflora. MTT assay of lipoidal matter for in vitro for cytotoxic activity towards HCT-116 carcinoma cell line showed IC50 of 38.76 µg/mL. Using agar well diffusion, S. tomentosa displayed moderate antimicrobial activity at concentration of 50 mg/mL. (see constituents above) (13)
• Neuroprotective / Prevention of Induced Parkinsonism: Study evaluated the phytoconstituents, radical scavenging capacity, and neuroprotective mechanism against of ST extract against MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine)-induced Parkinsonism assaying the activities of the dopaminergic system and antioxidant defenses, glycogen synthase kinase 3ß (GSK3-ß) phosphorylation, and α-synuclein levels in C57BL/6 mice. In vitro, the extract showed high ABTS radical scavenging capacities through higher phenolic contents, especially protocatechuic acid and epicatechin. Results showed potential of the extract to counteract MPTP-induced motor deficit. The neuroprotective mechanism against MPTP-induced parkinsonism might be related to decreasing GSK-3ß phosphorylation and restoring the activities of striatal antioxidant defenses to restore nigrostriatal dopaminergic function and decrease α-synuclein accumulation. (14)
• Anti-SARS-CoV-2 and Anti-MERS-CoV: Study evaluated the anti-MERS-CoV activities of three medicinal plants (Sophora tomentosa, Azadirachta indica, and Artemisia judaica). S. tomentosa recorded the highest viral inhibition percentage (96%), with virucidal main mode of action. The anti-SARS-CoV-2 activity was very high (100%). Phytochemical investigation identified nine compounds. Compound 4 (genistein 4'-methyl ether) showed superior anti-SARS-CoV-2 activity with IC50 of 2.13 µm, with very effective virucidal and adsorption mechanisms. IC50s against SARE-CoV-2 and MERS-CoV were 1,01 and 3.11 µg/mL, respectively. (16)
• Insecticidal against Culex pipiens / Quinolizidine Alkaloid: Study evaluated of Sophora tomentosa and S. secundiflora alkaloids fraction isolated one new quinolizidine alkaloid (1) 13-methoxyanagyrine together with six known ones (2-7). Study evaluated the insecticidal activity of 70% methanol extract of leaves of S. tomentosa and S. secundiflora and isolated alkaloids against 3rd instar larvae of Culex pipiens. Sophora tomentosa showed highest mortality rate with LC50 of 3.11 ppm after 24h and 0.66 ppm after 48h, and anagyrine (6) exhibited remarkable insecticidal activity with LC50 of 3.42 ppm after 24 h exposure. On cytotoxicity testing, anagyrine (6) exhibited IC50s of 25.3 and 30.2 µg/mL against MCF-7 and HEPG2 cancer cells, respectively. (18)
Availability
Wild-crafted. |

![]()



 Gen info
Gen info
 - Study of aerial parts of Sophora tomentosa isolated two new benzofuran derivatives (1), mp 235 - 2370, C15H10O5 and (2), mp 179 - 1810, C16H12O5, together with 1-maackiain, stigmasterol, medicagol, formononetin and 2',4',4-trihydroxychalone (isoliquiritigenin). (
- Study of aerial parts of Sophora tomentosa isolated two new benzofuran derivatives (1), mp 235 - 2370, C15H10O5 and (2), mp 179 - 1810, C16H12O5, together with 1-maackiain, stigmasterol, medicagol, formononetin and 2',4',4-trihydroxychalone (isoliquiritigenin). (
 Parts used
Parts used